

Photo(Electro)Catalysis Laboratory


Indian Institute of Technology Delhi
Photo(electro)catalytic sustainable conversions
Our group is focused on developing earth-abundant element-based inorganic materials, which include polyoxometalate-based novel compounds. We explore them for the photo(electro)catalytic conversions of small molecules like, H2O, CO2, [NO]3-, NH3 to their valuable counterparts. Further, we also explore their structure-function correlation to interpret the reactivity of the material and to establish the underlying reaction mechanism.
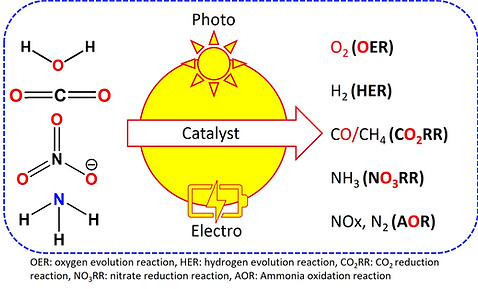
Hydrogen (H2) is now being used as alternative to carbonaceous fuel because of highest energy density and non-toxic waste generation. However, electrochemical water splitting which consists of two important reactions; hydrogen evolution reaction (HER) and oxygen evolution reaction (OER), is considered as one of the most promising approach to produce H2, a renewable fuel. An efficient and durable catalyst must operate both of the half cell reactions (HER and OER) simultaneously under low over-potentials and exhibit fast reaction kinetics. Our group is focused on the development of non-noble metal based bifunctional catalysts for overall water splitting as a economic and sustainable development towards a renewable energy based society.
Design of metal chalcogenides for water splitting



Chakraborty et al. JACSAu 2024, 4, 642-656
Chakraborty et al. J. Mat. Chem. A 2024, 12, 30810-30820
Chakraborty et al. Nanoscale, 2024, 16, 7467
Metal-sulfide
Wolframite-type metal oxide
Spinel
Design of polyoxometalate-stabilized metal oxide catalysts
Polyoxometalates (POMs) are early transition metal containing anionic inorganic clusters with variable dimensions and property depending on the metal ions present in it. Our group is involved in stabilizing various metal-oxide quantum dots utilizing POMs as the surface protecting ligands. Conjugation of POMs to the surface can control the of structure and function of these complete inorganic structures. We are applying these hybrid structures in various energy and/or photo(electro)catalytic conversions, like water splitting, CO2 reduction, nitrate reduction (NO3RR), and photochemical ammonia synthesis.

Chakraborty et al. ACS Appl. Mat. Interfaces 2024, 16, 32385
Electrosynthesis of NH3

Photochemical CO2 reduction
Chakraborty et al. Chem. Eur. J. 2024, 30,
e202400428
Electrochemical H2O splitting

Chakraborty et al. Chem. Eur. J. 2023, 29,
e202203033

Photosynthesis of NH3
Chakraborty et al. Small 2026, 22, e13455

Chakraborty et al. Inorg. Chem. 2026, 65, 4287
Electrochemical NO3RR
Electrochemical ammonia oxidation reaction (AOR)
Due to the low anodic potential of AOR, NH3 electrolysis has been considered to be a potential alternative to water electrolysis, where the conversion of NH3 into N2/NOx in the anode occurs at mild to low potential, and H2 can be produced from the cathode at a low cell potential. Our group is also exploring novel inorganic material as anode material for the AOR with a primary motif on product selectivity and reaction mechanism.

Chakraborty et al. J. Mat. Chem. A. 2026, 14, 9492
AOR on Zn-doped MnWO4

Chakraborty et al. ACS Applied Nano Mater. 2024, 7, 8329-8340
AOR on nickel foam

AOR Pt supported on NiWO4
Chakraborty et al. ACS Applied Materials & Interfaces, 2025, 17, 9391-9406
Phosphamide catalyzed organic transformation, proton and CO2 reduction

Phosphamide, structurally analogous to phosphine oxide, has never been explored for the above-mentioned phosphorous-mediated redox (PV/III) catalysis. Our group have designed a series of molecular phosphamide, polyphosphamides and phoapahmide on metal oxide supports as heterogeneous catalysts for the organic conversions like Appel or Wittig reactions. We are looking forward to design novel poly-phosphamides which will show potential photo(electro)catalytic conversion of small molecules into valuable organic and inorganic feedstocks.


Appel Reaction
CO2 reduction/utilization
Proton conduction/reduction
Chakraborty et al. J. Org. Chem. 2024, 89, 15851
Chakraborty et al. Small 2025, 21, 2412256
Chakraborty et al. J. Mat. Chem. A 2025, 13, 21480

Wittig Olefination Reaction
Chakraborty et al. Chem. Comm. 2025, 61, 16058
C=N condensation via HWE

Chakraborty et al. Inorg. Chem. 2026, (in revision)
International Recognitions
PEC Lab updates!
-
Congratulation to Dr. Anubha Rajput and Dr. Laxmikanta Mallick our two recent PhD graduates!
-
Congratulations Anup for his recent paper in J. Mat. Chem. A 2025!
-
Congratulations Hirak for his first research article as first author in ACS Appl. Mater. Interfaces 2025!
-
PEC lab welcomes Muskan, the new PhD student!
-
Congratulations Nidhi for her research article in Small 2025!
-
Congratulations Dikshita for her first research article in Small 2025!
-
Congratulations Anubha and Avinava for their J. Mat. Chem. A 2024 papers!
-
Congratulations Dr. Chakraborty for being promoted to Associate Professor!
-
PhD position available time to time! Candidates with M.Sc. in chemistry/inorganic chemistry background may apply through IITD application portal for June or December session each year.
-
Institute or personal funded postdoc positions are not available in the group. PEC lab can only host the externally funded postdocs. Interested postdoc candidate with PhD in inorganic chemistry may write application/proposal for NPDF (ANRF), CSIR-RA or any other funding opportunities may contact.







